|
18 Although most AA patients exhibit no signs or symptoms of PNH early in the natural history of their disease when the PNH clone size is small, many, but not all, will experience further expansion of the PIG-A mutant clone and progress to classic PNH. 13 DNA sequencing of the GPI-AP–deficient cells from AA patients reveals clonal PIG-A gene mutations. 16, 17 Typically, fewer than 10% GPIAP–deficient granulocytes are detected in AA patients at diagnosis, but occasional patients may have larger clones. Acquired AA is an autoimmune disorder, where the target of the immune attack is primitive CD34 + bone marrow progenitors. 13 – 15 In contrast to patients with classic PNH, these patients typically have a lower percentage of PNH cells. Hemoglobinuria, smooth muscle dystonias (eg, esophageal spasm and erectile dysfunction), severe fatigue, and thrombosis are common in patients with classic PNH.Īn expanded PNH clone is also found in up to 70% of patients with acquired AA demonstrating a pathophysiologic link between these disorders. 
These patients tend to have a normocellular to hypercellular bone marrow with erythroid hyperplasia, an elevated reticulocyte count, a large population of PNH cells (usually > 60% PNH granulocytes) and a lactic dehydrogenase (LDH) that is 2 to 10 times the upper limit of normal. Patients with classic PNH have signs and symptoms of intravascular hemolysis. Thrombosis is the leading cause of death, but others may die of complications of bone marrow failure, renal failure, myelodysplastic syndrome, and leukemia. 9 – 12 The median survival is 10 to 15 years, but with a wide range. The natural history of PNH is highly variable, ranging from indolent to life-threatening. Free plasma hemoglobin scavenges nitric oxide and depletion of nitric oxide at the tissue level contributes to numerous PNH manifestations, including esophageal spasm, male erectile dysfunction, renal insufficiency, and thrombosis. 8 Intravascular hemolysis releases free hemoglobin into the plasma. The loss of complement regulatory proteins renders PNH erythrocytes susceptible to both intravascular and extravascular hemolysis, but it is the intravascular hemolysis that contributes to much of the morbidity and mortality from the disease. 6, 7 CD55 inhibits C3 convertases and CD59 blocks formation of the membrane attack complex (MAC) by inhibiting incorporation of C9 into the MAC. Two of these proteins, CD55 and CD59, are complement regulatory proteins the absence of these proteins is fundamental to the pathophysiology of the disease. Consequently, the PNH stem cell and all of its progeny have a reduction or absence of glycosyl phosphatidylinositol (GPI)–anchored proteins. 
The PIG-A gene product is required for the biosynthesis of glycophosphatidylinositol anchors, a glycolipid moiety that attaches dozens of proteins to the plasma membrane of cells. 4, 5 Expansion and differentiation of the PIG-A mutant stem cell lead to clinical manifestations of the disease. 3 The disease originates from a multipotent hematopoietic stem cell that acquires a mutation of the PIG-A gene. 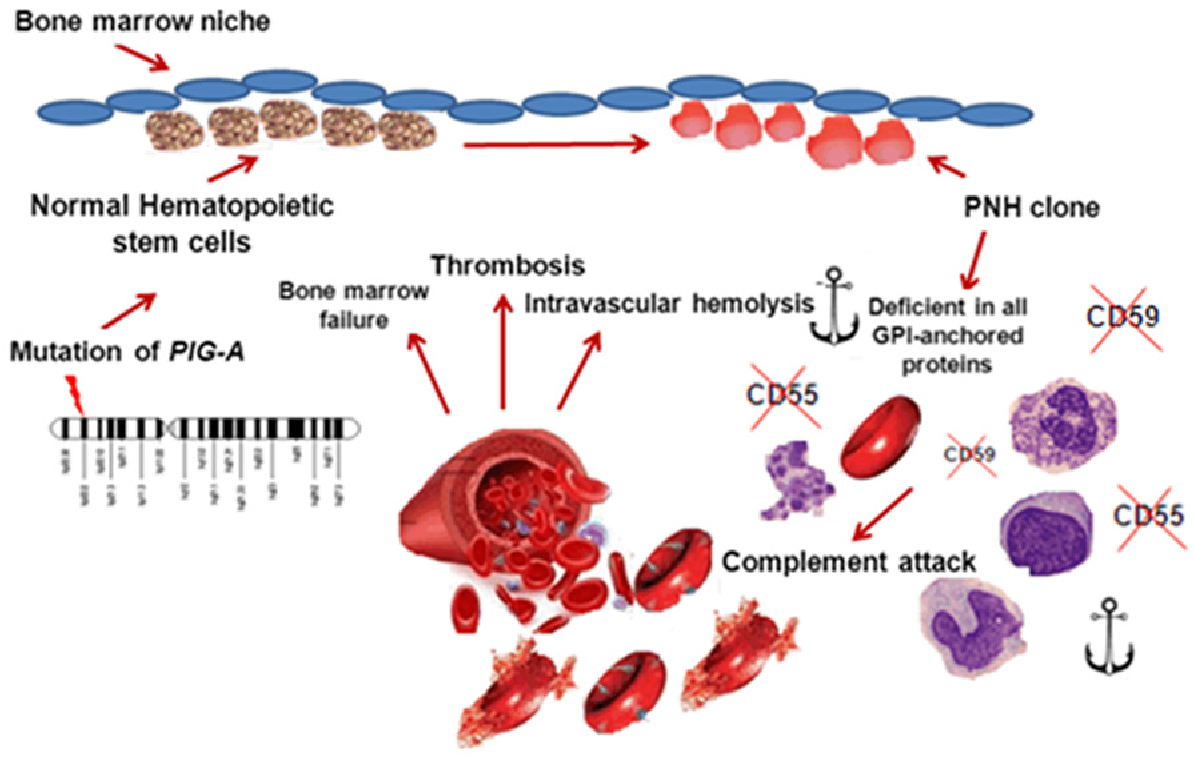
1, 2 PNH can arise de novo or in the setting of aplastic anemia (AA). Paroxysmal nocturnal hemoglobinuria (PNH) is a clonal hematopoietic stem cell disease that can present with bone marrow failure, hemolytic anemia, smooth muscle dystonias, and thrombosis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
